Abstract of Publication No. 532
 Gaël Labat, Colette Boskovic and Hans U. Güdel
Gaël Labat, Colette Boskovic and Hans U. Güdel
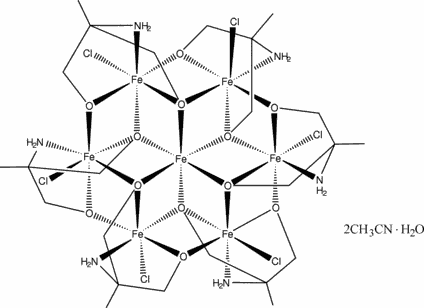
Hexakis(m4-2-amino-2-methylpropane-1,3-diolato)hexachloroheptairon(II,III) acetonitrile disolvate monohydrate
Acta Cryst. E 61, m611-m613 (2005)
![]()
![]()
![]()
Abstract:
Single crystals of a new mixed-valence compound,
[Fe7(C4H9NO2)6Cl6]·2CH3CN·H2O, were
synthesized by solvothermal methods. The crystal structure consists of discrete
heptanuclear complexes which are linked by weak intermolecular C-H···Cl hydrogen bonds to form a
two-dimensional network. In each complex, a central Fe atom is connected to six
surrounding Fe centres by alkoxo-type O atoms from the organic ligand.
Bond-valence sum calculations indicate that the central Fe ion is
FeII, as are two of the outer Fe centers, while the
remaining four Fe centers are FeIII. Although
the outer Fe atoms are crystallographically equivalent, it is uncertain whether
this arises from positional disorder or charge delocalization. The molecular
structure has S6 point symmetry.